Eli lilly
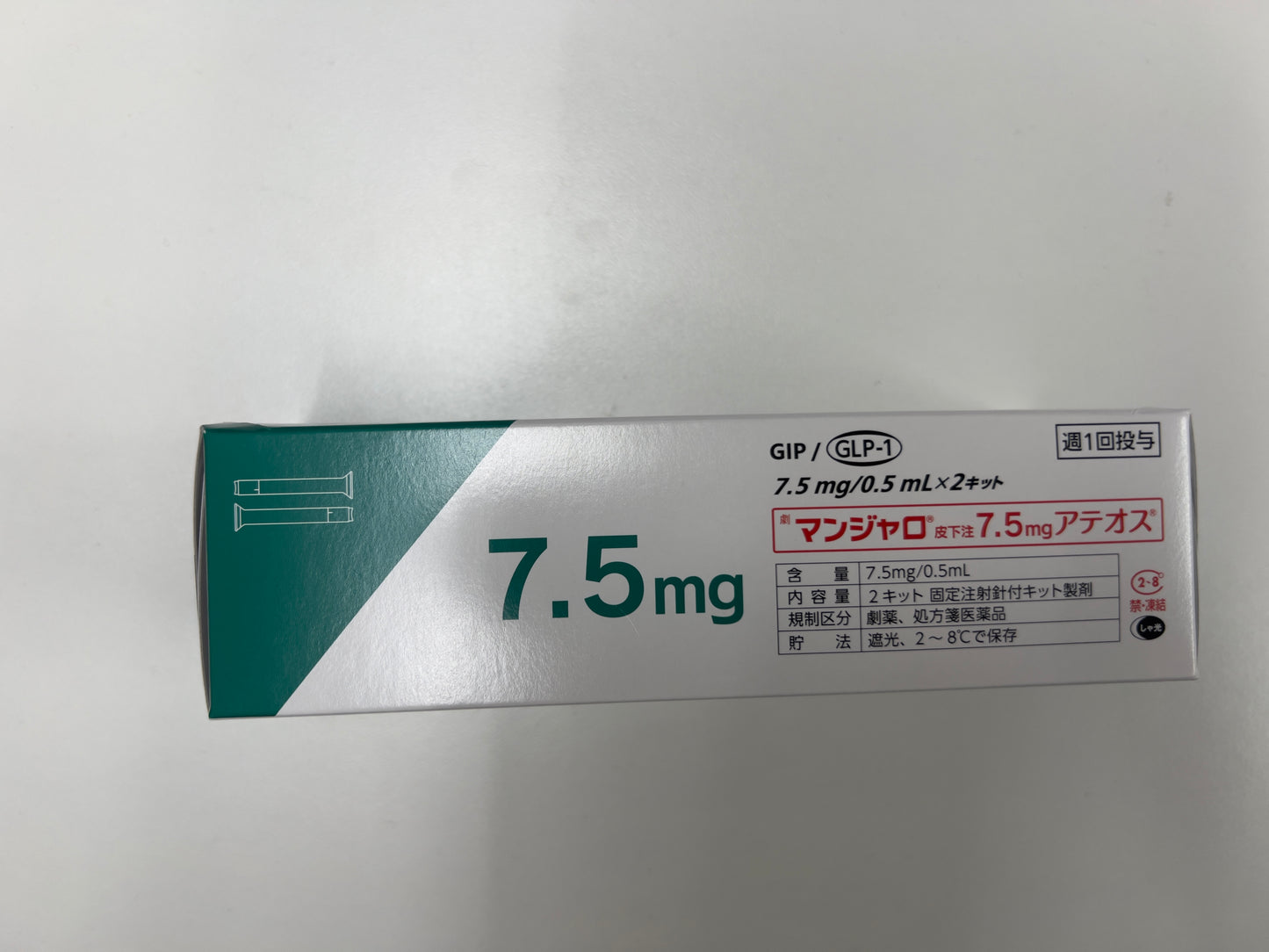
Tirzepatide Injection 7.5mg
Tirzepatide Injection 7.5mg
Couldn't load pickup availability
Key Selling Points of Tirzepatide for Diabetes and Obesity Management
1. Innovative Dual Receptor Mechanism
• First-in-Class Dual Agonist: Tirzepatide is the world’s first dual agonist of glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptors.
◦ GLP-1 Receptor: Delays gastric emptying, inhibits glucagon secretion, and stimulates insulin release to lower blood sugar.
◦ GIP Receptor: Enhances insulin sensitivity, promotes fat metabolism, and increases energy expenditure, synergizing with GLP-1 for superior metabolic control.
• Precision Metabolic Regulation: Mimics natural GIP/GLP-1 pathways to simultaneously improve glycemic control, reduce weight, and optimize lipid/energy balance.
2. Clinically Proven Efficacy
• Exceptional Blood Sugar Control:
◦ Reduces HbA1c by 1.5%–2.0% in type 2 diabetes patients, outperforming many single-receptor therapies.
◦ Helps patients achieve glycemic targets faster, even when prior treatments fail.
• Powerful Weight Loss:
◦ Leads to 15% or more weight reduction in obese/overweight patients in long-term studies, with sustained effects.
◦ Suppresses appetite and increases satiety, addressing the root cause of overeating.
• Cardiovascular & Metabolic Benefits:
◦ Lowers triglycerides, LDL cholesterol, and blood pressure, reducing cardiovascular risk.
3. Convenient and Patient-Friendly Administration
Function and Indications
Indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus by mimicking the action of natural GIP and GLP-1, promoting insulin secretion, inhibiting glucagon release, and delaying gastric emptying.
Applicable Population
• Primary Use: Adults with type 2 diabetes who have inadequate glycemic control through lifestyle modifications alone.
• Additional Use: Adults with obesity (BMI ≥28 kg/m²) or overweight (BMI ≥24 kg/m²) and at least one weight-related comorbidity (e.g., hypertension, dyslipidemia, obstructive sleep apnea), for weight management.
Contraindications
• Personal or family history of medullary thyroid carcinoma (MTC) or multiple endocrine neoplasia syndrome type 2 (MEN 2).
• Hypersensitivity to tirzepatide or any excipients.
Usage and Dosage
• Starting Dose: 2.5 mg once weekly, administered subcutaneously.
• Dose Titration: After 4 weeks, increase to 5 mg/week; may further increase by 2.5 mg/week (to a maximum of 15 mg/week) based on glycemic control or weight loss response.
• Administration: Inject into the abdomen, thigh, or upper arm; rotate injection sites to avoid skin reactions.
Adverse Reactions
• Common (≥5%): Nausea, vomiting, diarrhea, abdominal pain, constipation, headache, fatigue.
• Serious Risks:
◦ Thyroid C-cell tumors: Observed in animal studies; contraindicated in patients with MTC history/family history.
◦ Hypoglycemia: Increased risk when combined with insulin secretagogues (e.g., sulfonylureas) or insulin.
Share